Craze Assessment —Data from the plan microbial environmental monitoring program that can be linked to time, change, facility, and so forth. This facts is periodically evaluated to ascertain the standing or pattern of that application to verify whether it's below adequate Manage.
The techniques useful for identification of isolates need to be verified making use of indicator microorganisms (see
On the other hand, it has been suggested that when the isolator is in a very managed surroundings, the potential for contaminated item is minimized while in the celebration of the pinhole leak in the match or glove.
FARRAR® has two diverse methods to practical experience our items. At our headquarters in Davidson, NC, our BioSolutions House is made up of fully operational ULC units with regular product dealing with selections - Be happy to visit this Room to strategy your challenge and perform with our design workforce with a tailored materials managing Answer that fits your approach.
Adverse Strain. To keep the circulation of contamination in a single direction, air is pulled out of the room and straight exhausted outside. Once more, due to the fact design focuses on basic safety, it is vital that Innovative mechanical methods regularly pull air out of the room.
Documentation and Validation: Each and every step of the cleanroom operation, from environmental checking to cleaning strategies, need to be documented to be sure compliance with regulatory standards and provide traceability through inspections or audits.
4. A favourable really should be taken care of in the area to prevent the doorway of particles and contamination through the air.
The first location where by the Uncooked and packaging supplies are exposed to the room setting is in the incoming sampling room. Listed here containers need to be opened to choose samples of the Uncooked and packaging components. The requirements for this area have to be the same as during the production click here spot which is typically ISO eight Course a hundred,000. The changeover position amongst a warehouse as well as the production location is definitely the dispensary or weigh room.
Microbiological monitoring of personnel can be staying included into education systems. Normal testing of gloves and gowns utilizing Speak to plates or swabs delivers fast feed-back over the success of aseptic procedures and can help reinforce great tactics.
These controlled environments should adjust to rules which means that sanitation should be fulfilled. Fumigation serves as a vital decontamination procedure. The report testimonials the validation of fumigation procedures in pharmaceutical cleanrooms and discusses in detail the fumigation system and its difficulties along with their compliance.
Generally, check here as soon as the validation establishes the performance in the barrier method, the frequency of sampling to observe the microbiological standing from the aseptic processing area can be diminished, when compared to the frequency of sampling of classical aseptic processing techniques.
Sterilization and Decontamination in Cleanrooms – Pharmaceutical Technological innovation – This resource supplies an summary of sterilization and decontamination methods in cleanrooms, concentrating on solutions like ethylene oxide, hydrogen peroxide, and moist heat sterilization, and their worth in sustaining a sterile natural environment.
Every classification serves a certain objective, guaranteeing that the level of contamination control matches the producing phase’s needs.
The modular construction of your EcoFlex permits the products to be redeployed following room modifications or relocation.
 Barret Oliver Then & Now!
Barret Oliver Then & Now!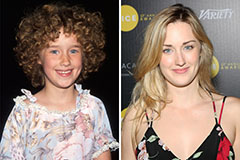 Ashley Johnson Then & Now!
Ashley Johnson Then & Now!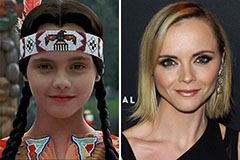 Christina Ricci Then & Now!
Christina Ricci Then & Now!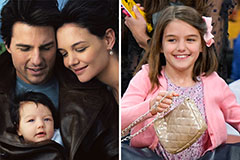 Suri Cruise Then & Now!
Suri Cruise Then & Now! Daryl Hannah Then & Now!
Daryl Hannah Then & Now!